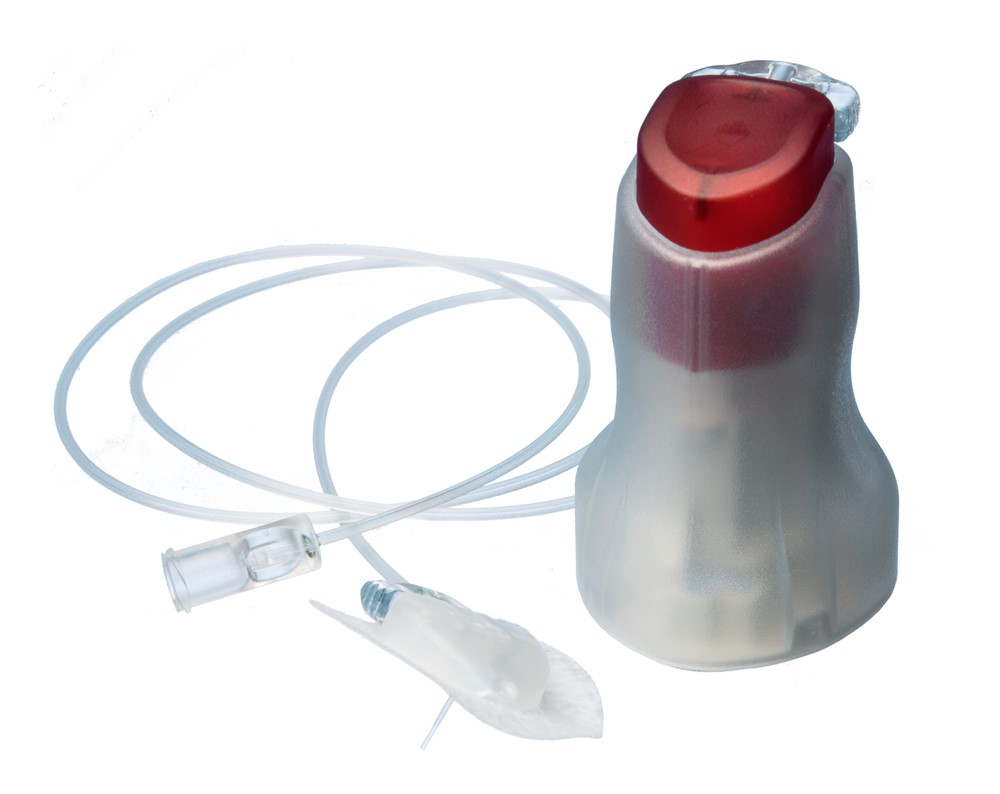
ConvaTec and neria™ Infusion Sets
ConvaTec Infusion Care specializes in manufacturing subcutaneous infusion sets since the 1980s. Each year, we provide patients with millions of sets for subcutaneous drug delivery.
ConvaTec Infusion Care is the world’s largest manufacturer of subcutaneous infusion sets. Our success is based on a high degree of innovation supported by user-friendly design and superior quality.
All over the world, ConvaTec infusion sets improve the quality of life and treatment options for users. These are widely used for subcutaneous infusion treatment areas, such as Diabetes, Parkinson’s Disease, Primary Immune Deficiency, Pain Management and more.
We closely follow developments within subcutaneous drugs to identify any new treatments where our user-friendly and safe solutions could be advantageous for patients.
If you are interested in hearing more about our capabilities, please visit: www.b2b.convatec.com.
ConvaTec Infusion Care is a franchise of ConvaTec Plc. and is located in Denmark.

High level of Quality
To realise our vision - pioneering trusted medical solutions to improve the lives we touch - and generate value, we must build trust and develop positive stakeholder relationships. We endeavour to do this by running our business in a responsible way.
ConvaTec Infusion Care manufactures neria™ infusion sets according to the highest regulatory standards. Our sets do not contain latex or PVC1,2 and are designed for safe and intuitive use by complying with EU, US and WHO guidelines and regulations on safer sharps.3,4,5
All of our products, processes, and production sites live up to a high level of quality that is maintained professionally and efficiently through our quality management system.
Our production sites are certified and approved by regulatory agencies across the world. This guarantees that our product lives up to the required standards and ensures a high level of safety for clients and product users.
For more details on our approach to corporate responsibility and sustainability, please visit our group site.
1. Statement - Latex Statement-LXUE23OCT2020. Data on file. Unomedical A/S.. 2. Statement - PVC Statement-LXUE23OCT2020, Data on file. Unomedical A/S. 3. COUNCIL DIRECTIVE 2010/32/EU of 10 May 2010. 4. Needlestick Safety and Prevention Act (NSPA), 5. World Health Organization. WHO Guideline On The Use of Safety-Engineered syringes for Intramuscular, Intradermal and Subcutaneous Injections In Health-Care Settings. Geneva, Switzerland: World Health Organization; 2016.
neria™ Infusion Sets and Needle Safety
At ConvaTec Infusion Care, the safety of both patients and healthcare professionals is our highest priority. One of our key focus area is helping to prevent needle stick injuries, which are shown to have negative consequences both economically and psychologically. With our latest product development - neria™ guard infusion set - we aim to eliminate this risk.
The neria™ guard infusion set is the only commercially available infusion set in the world with a fully automatic insertion technique that is safe and has a hidden needle. The neria™ guard infusion set meets the highest standards for needle safety regulations1,2,3 and may help reduce the clinical, economic and social burden associated with needle stick injuries.4
1. COUNCIL DIRECTIVE 2010/32/EU of 10 May 2010, 2. Needlestick Safety and Prevention Act (NSPA), 3. World Health Organization. WHO Guideline On The Use of Safety-Engineered syringes for Intramuscular, Intradermal and Subcutaneous Injections In Health-Care Settings. Geneva, Switzerland: World Health Organization; 2016. 4.Costigliola V, Frid A, Letondeur C, Strauss K. Needlestick injuries in European nurses
in diabetes. Diabetes Metab. 2012;38(Suppl 1):S9–S14.